Serving nanoparticle "soup"
A research collaboration involving groups in Regensburg, Freiburg, Perth, and Glasgow has shown [1] that the strange properties characteristic of room temperature ionic liquids (RTILs) are due to the presence of a high degree of structure. RTILs are the subject of intensive research as a promising new breed of green solvents for industry with low toxicity and lower environmental impact than traditional organic solvents. RTILs consist entirely of molecular ions and have exceptionally low volatility but also have properties that can be fine-tuned – through changes in the molecular structure – to provide more efficient routes of synthesis. However, the physical properties of RTILs are poorly understood; for example, they are extremely viscous but it is not understood why this is so.
The study found that RTILs organise themselves spontaneously into fluid nanoparticles floating in a liquid sea. This nanoparticle “soup” acquires greater viscosity much like mayonnaise is more viscous than its constituents, oil and vinegar. The physics behind this effect was developed by Einstein in the early 20th century.
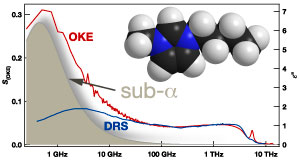
The discovery was made possible by terahertz spectroscopy, which is an exciting new technology that has also been used for medical imaging and weapons detection. Terahertz spectroscopy bridges the gap between microwaves and infrared radiation where materials absorb radiation characteristic of their structure.
The collaborating groups have combined four types of terahertz spectroscopy to produce the detailed picture necessary to reveal this structure. At Strathclyde, pulses of infrared light shorter than a millionth of a millionth of a second were used to induce and then probe fluctuations in the refractive index that are determined by the microscopic liquid interactions. These interactions in turn determine the macroscopic properties: viscosity, density, thermal conductivity, and freezing and boiling points, that are essential to their applications.
For more information contact Klaas Wynne ( klaas.wynne ![]() phys.strath.ac.uk) or Prof Richard Buchner (richard.buchner
phys.strath.ac.uk) or Prof Richard Buchner (richard.buchner ![]() chemie.uni-regensburg.de).
chemie.uni-regensburg.de).
[1] David A. Turton et al., J. Am. Chem. Soc. 131, 11140-11146 (2009) DOI: 10.1021/ja903315v. (local copy of pdf)
What are room-temperature ionic liquids?
A typical ionic substance is a salt such as NaCl (sodium chloride), which – when heated to 801° C – will melt to form a liquid consisting of sodium cations and chloride anions. Because of the strong Coulomb attraction between ions, such a liquid has a very low vapour pressure and will not evaporate and therefore might be an ideal solvent for industrial-scale synthesis. However, the high temperature is clearly an obstacle for organic chemistry as most organic compounds would decompose. Room temperature ionic liquids (RTILs) typically consist of rather large organic cations and smaller inorganic anions. For example, the RTILs used in the study described here have 1,3-dialkylimidazolium-based cations. Such RTILs are liquid around room temperature while having the same low vapour pressure as inorganic salts such as NaCl. This allows chemists to perform chemical reactions in a polar liquid with relatively easy recovery of the solvent afterwards.
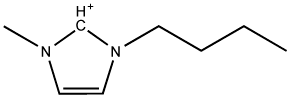
The molecule 1-N-butyl-3-methylimidazolium (bmim) is a commonly used organic cation in room-temperature ionic liquids (RTILs).
What is terahertz spectroscopy?
Terahertz spectroscopy is spectroscopy at frequencies of ~1 THz = 1012 Hz. Radiation with a frequency of 1 THz has a wavelength of 0.3 mm and therefore lies between the microwave and the infrared region. The collaboration combines microwave spectroscopy with time-domain terahertz spectroscopy and Fourier-transform infrared spectroscopy to cover a range from sub-GHz to 20 THz. In addition, optical Kerr-effect spectroscopy (a form of Raman spectroscopy) is used to obtain a continuous spectrum from sub-GHz to 30 THz. Such spectra can measure slow diffusive motions (related to viscosity) in the GHz range, through cage rattling motions in the low THz range, to vibrations in the high THz range.
Acknowledgements
We thank W. Kunz and H. Helm for laboratory facilities at Regensburg and Freiburg and acknowledge funding from the U.K. Engineering and Physical Sciences Research Council (EPSRC) and the Deutsche Forschungsgemeinschaft within Priority Program 1191.